ArchivesCategories |
Back to Blog
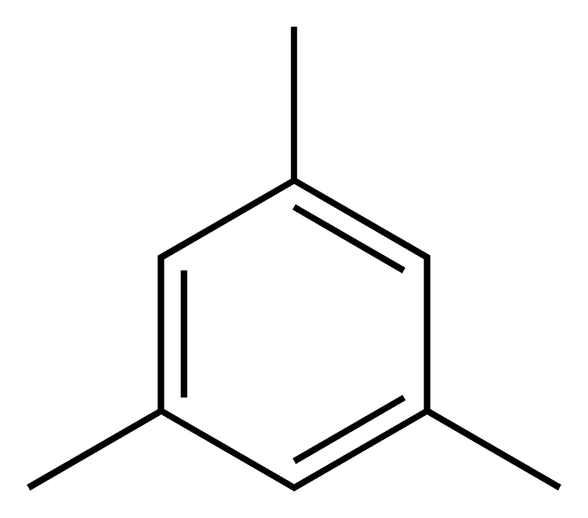
Mesitylene, commonly known as 1, 3, 5-trimethylbenzene, has the chemical formula C6H3 (CH3)3. It is an odourless liquid with a recognisable fragrant scent. Mesitylene belongs to the group of substances known as aromatic hydrocarbons, which are distinguished by their strong fragrance and cyclic arrangement of carbon atoms.
By alkylating benzene with propylene, mesitylene is made commercially. Small quantities of it are also naturally present in some kinds of crude oil and coal tar. The melting point of mesitylene is -45°C and its boiling point is 166°C. Although it is insoluble in water, several organic solvents, including benzene, toluene, and chloroform, can dissolve it. Mesitylene has a high degree of symmetry, which is one of its most prominent characteristics. With regard to its three methyl groups and benzene ring, the molecule is symmetrical. Mesitylene has several intriguing physical characteristics as a result of its symmetry, such as the capacity to crystallise with a high degree of order. Mesitylene is frequently employed as a solvent in organic chemistry, especially in processes involving delicate substances or high temperatures. It is a great option for high-temperature reactions due to its low vapour pressure and comparatively high boiling point. Mesitylene is also utilised as a raw material to make other organic compounds, such medications and colours. Mesitylene is used as a fundamental component in the synthesis of metal-organic frameworks (MOFs), which is a fascinating application. A type of materials known as MOFs is made up of metal ions or clusters that are joined by organic ligands. These substances have a broad variety of possible uses, such as gas storage, sensors, and catalysts. Because of its capacity to bind with metal ions, mesitylene can be employed as a ligand in the production of MOFs. Mesitylene may be used as a gasoline additive, according to other research. Gasoline's octane rating, a gauge of a fuel's capacity to withstand knocking or explosion, can be raised by adding mesitylene. Increasing the octane rating is a crucial objective for petrol makers since knocking may result in engine damage and decreased performance. Mesitylene is an appealing option for a gasoline additive since it is a very inexpensive and plentiful substance. Despite having several beneficial uses, mesitylene does have some disadvantages. Its potential toxicity as well as its flammability make it a dangerous substance. Mesitylene exposure can irritate the skin, eyes, and respiratory system. Although additional study is required to properly grasp its possible health consequences, it is also thought to be a probable carcinogen. Mesitylene is a valuable and adaptable substance having several uses in organic chemistry, materials science, and fuel technology. It is a desirable option for a variety of applications due to its high degree of symmetry and comparatively low toxicity. To guarantee its safe usage, mesitylene must be handled carefully and cautiously, just like any other chemicals.
0 Comments
Read More
Back to Blog
Modern medicine would not be possible without the disposable asepsis-safe injection needle. Patients are given medications and vaccinations with the help of this device, and fluids are also extracted for diagnostic purposes. The needle is made to only be used once, lowering the danger of infection and patient-to-patient transmission. The significance of disposable asepsis-safe injection needles and the advantages they provide will be covered in this article.
The materials used to construct disposable asepsis-safe injection needles are of the highest calibre and are intended to be sterile and non-toxic. Strict quality control procedures are used during the manufacturing process to guarantee the safety and efficacy of the needles. Because the needle should only be used once, there is less chance of infection and contamination. Additionally, the needle is made to be simple to use, making it perfect for medical personnel who need to provide injections swiftly and precisely. The fact that disposable asepsis-safe injection needles are made to be sterile is one of their main advantages. Because of this, they are devoid of viruses, germs, and other dangerous microbes that might infect people. By using heat, radiation, or chemicals to destroy every microbe on the needle's surface, sterilisation is accomplished. The needle is put in a sterile container once it has been sterilised to keep it safe from contamination until it is time to use it. The convenience of using disposable asepsis-safe injection needles is another advantage. It is simple to puncture the skin and give medication or extract fluids thanks to the needle's sharp, accurate shape. The likelihood of pain or discomfort during the injection procedure is decreased since the needle is also made to be pleasant for the patient. Disposable asepsis Safe injection needles are also advantageous since they lower the possibility of patient cross-contamination. Reusing needles runs the danger of transmitting bacteria from one patient to another, which can result in infections and other health issues. Medical personnel may avoid this danger and guarantee that every patient receives a clean injection by utilising disposable needles. Disposable asepsis safe injection needles offer these advantages in addition to being economical. Disposable needles may initially cost more than reusable ones, but they save a lot of money over time. If needles are reused, they must be sterilised after every use, which adds more time, money, and equipment requirements. Disposable needles allow medical personnel to skip the sterilisation step and cut down on the time and materials needed for each injection. A disposable asepsis to stop the transmission of dangerous illnesses, it is crucial to have safe injection needles. Reusing needles increases the possibility of transmitting infectious diseases including HIV, hepatitis, and other blood-borne illnesses from one patient to another. Medical personnel may avoid this danger and guarantee that every patient receives a secure and sanitary injection by utilising disposable needles.
Back to Blog
The detection and measurement of certain proteins or antibodies in biological samples is made possible by the use of an immunofluorescence analyzer, a potent diagnostic tool utilised in clinical laboratories and medical research. For the purpose of locating and quantifying the presence of certain biomolecules inside cells or tissues, it combines immunology and fluorescence microscopy methods.
An immunofluorescence analyzer works by using fluorescently-labeled antibodies that attach to the target molecules selectively. Animals that have been exposed to the target antigens and the resulting immunological response are used to manufacture these antibodies. Following their purification, the antibodies are coupled with fluorescent dyes like FITC or rhodamine. The fluorescently-labeled antibodies are then treated with the biological material to be examined after it has been fixed onto a microscope slide. Any extra antibodies are washed away once the antibodies attach directly to the target molecules. A fluorescence microscope is used to see the material after it has been excited by light of a particular wavelength to reveal the fluorescent dye on the attached antibodies. As a result of the dye's longer wavelength light emission, the target molecules' position and intensity may be seen in an image produced by the microscope. Numerous medical illnesses, such as cancer, autoimmune diseases, and infectious diseases, are studied and diagnosed using immunofluorescence analyzers. An immunofluorescence analyzer, for instance, may find and measure the presence of viral antigens in biological samples like blood, urine, or sputum for diagnosing infectious disorders like viral infections. Utilising certain antibodies that bind to viral antigens enables the identification and measurement of the virus in the sample. Similar to this, an immunofluorescence analyzer may identify and measure the presence of autoantibodies that attack the body's own tissues in the diagnosis of autoimmune illnesses like rheumatoid arthritis. This makes it possible to identify and diagnose the illness early, which can help in the creation of effective treatment strategies. The expression of certain proteins or biomarkers linked to the onset and course of cancer can be found and quantified using immunofluorescence analyzers in cancer research. This can help with both the diagnosis and stage of the illness as well as the creation of specialised treatments. The excellent sensitivity and specificity of immunofluorescence analyzers make them an important tool for clinical diagnosis and medical research. To provide precise and repeatable findings, they call for meticulous sample preparation and handling. The accuracy and dependability of the results can be impacted by a number of variables, including the quality and specificity of the utilised antibodies, the incubation period, and the microscope settings. An immunofluorescence analyzer is a powerful diagnostic tool that combines immunology and fluorescence microscopy techniques to detect and quantify specific proteins or antibodies in biological samples. Its wide range of applications in medical research and clinical diagnosis makes it an invaluable tool for advancing our understanding of various diseases and developing targeted treatments. However, careful sample preparation and handling are essential to ensure accurate and reliable results.
Back to Blog
Cathepsins are a class of proteases that are active in a wide range of physiological and pathological processes. Cathepsin inhibitors are a class of drugs that suppress the action of cathepsins. Cathepsins, a kind of protease that breaks down and recycles cellular waste, are particularly crucial in lysosomes, the cellular organelles that break down and split protein molecules into smaller fragments.
There are many different types of cathepsins, but the most well-known are cathepsin B, L, and S. These enzymes are involved in a range of physiological processes, including protein turnover, antigen processing, and bone resorption. However, they are also implicated in a number of pathologies, including cancer, arthritis, osteoporosis, and cardiovascular disease. For a number of these disorders, cathepsin inhibitors have been looked into as prospective therapies. certain inhibitors may be able to reduce or stop the course of illness by obstructing the action of certain enzymes. Cathepsin inhibitors come in a variety of forms, such as peptidyl, vinyl sulfone, diazomethyl ketone, and epoxide inhibitors. Small peptides called peptidyl inhibitors resemble the cathepsins' organic substrates. Usually, they have an amino acid residue that binds to the enzyme's active site and stops the enzyme from cleaving its normal substrates. These inhibitors have a limited bioavailability and are quickly broken down by proteases in vivo, while being efficient in inhibiting cathepsin activity in vitro. Vinyl sulfone inhibitors are small molecules that contain a vinyl sulfone group, which irreversibly binds to the active site of the enzyme. These inhibitors are highly effective at blocking the activity of cathepsins in vitro and in vivo and have shown promise as potential therapeutics for a range of diseases, including cancer, arthritis, and osteoporosis. Diazomethyl ketone inhibitors are another class of small molecules that irreversibly bind to the active site of cathepsins. These inhibitors are highly effective at blocking the activity of cathepsins in vitro and in vivo and have shown promise as potential therapeutics for a range of diseases, including cancer, arthritis, and osteoporosis. Epoxide inhibitors are small molecules that contain an epoxide ring, which reacts with the active site of the enzyme. These inhibitors are highly effective at blocking the activity of cathepsins in vitro and in vivo and have shown promise as potential therapeutics for a range of diseases, including cancer, arthritis, and osteoporosis. Cathepsin inhibitors have been investigated as potential therapeutics for a number of diseases, including cancer, arthritis, and osteoporosis. In cancer, cathepsins are involved in tumor invasion and metastasis, making them attractive targets for therapy. In arthritis and osteoporosis, cathepsins are involved in bone resorption, making cathepsin inhibitors potential treatments to slow or prevent bone loss. Several cathepsin inhibitors are currently in clinical development for a range of diseases. For example, odanacatib, a selective cathepsin K inhibitor, is currently in clinical development for the treatment of osteoporosis. Talabostat, a dipeptidyl peptidase inhibitor that also inhibits cathepsin B, is in clinical development for the treatment of cancer.
Back to Blog
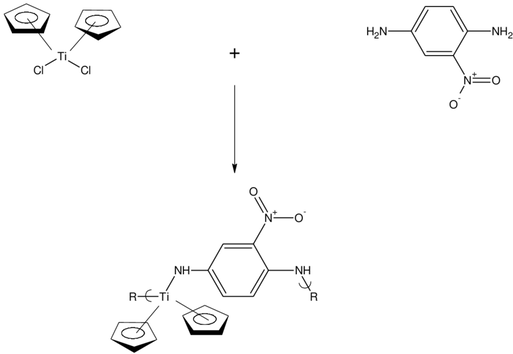
The chemical compound titanocene dichloride (TDC) has the formula (Cp2TiCl2), where Cp stands for the cyclopentadienyl ring. It is a solid that is a vivid orange colour and belongs to the family of compounds known as metallocenes, which are prized for their intriguing characteristics and usefulness in a variety of industries.
Geoffrey Wilkinson and F. Albert Cotton, two researchers who were examining the molecular characteristics of metallocenes, created titanocene dichloride for the first time in 1954. Since then, it has received extensive research due to its intriguing characteristics and possible uses. Being able to operate as a single-electron transfer agent, or being able to transfer one electron to another molecule or chemical, is one of TDC's most important features. As a result, it plays a crucial role in the field of organic synthesis where it is employed to start radical processes like polymerization. The capacity of TDC to form stable complexes with a variety of chemical molecules, including alkenes and alkynes, is another intriguing characteristic. For example, the creation of carbon-carbon bonds, these complexes have been investigated for their possible application as catalysts in organic processes. The possibility of using TDC as a cancer therapy has also been investigated. It has been discovered to be efficient against some cancer cell types and has been demonstrated to cause cell death in these cells. This is considered to be because TDC may interact with DNA, which can cause programmed cell death to be activated. TDC has also been investigated for its possible use in materials science, in addition to its prospective uses in organic synthesis and cancer therapy. It has been discovered to be a strong contender for the creation of conducting polymers, which are essential to the development of electrical devices like solar cells and LEDs. TDC has various drawbacks in addition to its potential uses. It is a highly reactive substance that, if handled improperly, might be toxic. Additionally, the cost of production prevents its broad usage. Titanocene dichloride is an intriguing substance that has received a lot of attention due to its unique features and prospective use in a variety of industries. It is a potential molecule for organic synthesis and cancer therapy due to its capacity to function as a single-electron transfer agent, form stable complexes, and cause cell death in cancer cells. It's a fascinating molecule for the creation of conducting polymers because of its prospective applications in materials science. But in order for it to be extensively employed in these applications, it must first be overcome because of its high reactivity, toxicity, and expense.
Back to Blog
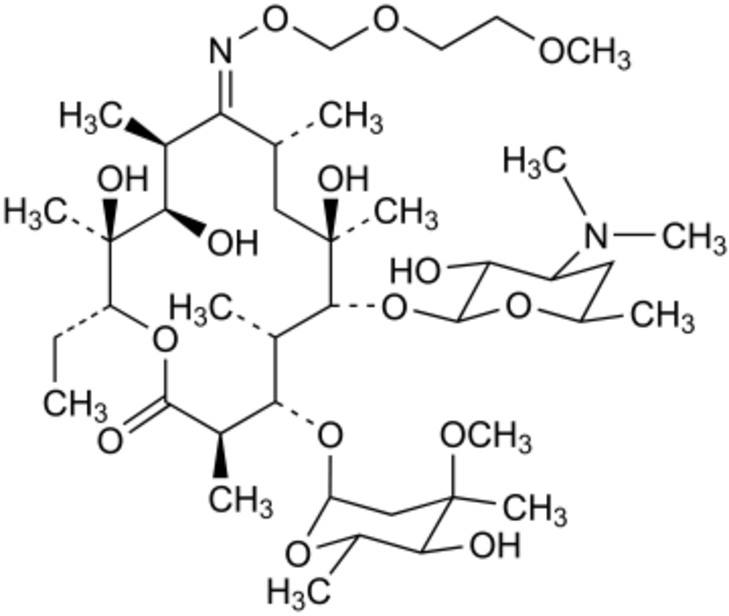
The macrolide category includes the antibiotic roxithromycin. It is used to treat bacterial infections such as those of the skin, urinary tract, and respiratory tract. Roxithromycin inhibits the production of the proteins that the bacteria need to thrive and survive.
Roxithromycin has been utilised in therapeutic settings since its discovery in the 1980s. It comes in a variety of forms, including pills, capsules, and suspensions. The drug normally has a low frequency of adverse effects and is well tolerated. It is a popular option for treating different bacterial illnesses because it is also reasonably priced. Roxithromycin has a broad-spectrum of activity against a variety of bacteria. It is effective against Gram-positive bacteria such as Streptococcus pneumoniae, Streptococcus pyogenes, and Staphylococcus aureus. It is also active against Gram-negative bacteria such as Haemophilus influenzae and Moraxella catarrhalis. Infections in the respiratory tract, such as bronchitis and pneumonia, are frequently treated with roxithromycin. Additionally, it is used to treat urinary tract infections including cystitis and pyelonephritis as well as skin infections like impetigo and cellulitis. Roxithromycin is also occasionally used to both prevent and treat infections in those with compromised immune systems, such as HIV-positive individuals. Roxithromycin might have negative effects, much like any antibiotic. Nausea, vomiting, diarrhoea, and stomach discomfort are the most typical adverse reactions. Most of the time, these side effects are minor and disappear on their own in a few days. Rarely, Roxithromycin may result in more severe adverse effects include hearing loss, allergic reactions, and liver damage. Any adverse effects must be reported right once to a healthcare professional. Roxithromycin is typically safe to use, although there are certain persons who shouldn't. Roxithromycin dose may need to be changed for those with liver or renal impairment, or they may need to stop taking it completely. Roxithromycin should not be taken by pregnant or nursing women unless absolutely required. Bacterial resistance building is one potential problem with Roxithromycin. Like other antibiotics, Roxithromycin can become less effective over time if it is misused or overused, which can result in the emergence of germs that are resistant to it. It's crucial to take Roxithromycin precisely as directed by a healthcare professional and to use it solely for bacterial infections if you want to avoid this from happening. Roxithromycin is a potent antibiotic that may be used to a number of bacterial illnesses. It is widely tolerated and has a wide range of activities. But it can have negative side effects and contribute to the emergence of bacterial resistance, just like any other antibiotic. Use Roxithromycin only as directed by a healthcare professional, and report any adverse effects right away to ensure its safety and effectiveness.
Back to Blog
In surgical operations, sterile X-ray detectable lap sponges are used as medical instruments to stop bleeding, clean wounds, and absorb fluids. Depending on the precise operation being carried out, these sponges can be found in a variety of sizes and forms and are normally manufactured from cotton or a cotton and rayon combination. Barium sulphate, a radiopaque substance that makes sponges visible on x-rays, is what distinguishes sterile x-ray detectable lap sponges from other sponges.
Using clean, x-ray detectable lap sponges during surgical operations is essential for guaranteeing patient safety. After surgery, if a sponge is still within the patient, it might result in life-threatening problems like infection, discomfort, and even death. By making them visible on x-ray imaging, x-ray detectable sponges lower the chance of retained sponges. This makes sure that any sponges unintentionally left inside the patient may be found and removed right away. Medical workers can also benefit from using sterile x-ray detectable lap sponges during surgical operations. The sponges are less likely to be accidentally moved or lost during surgery because to the radiopaque material's ability to help personnel identify them quickly. This is especially crucial for lengthier surgical operations that can include the use of multiple sponges. The production of sterile, x-ray detectable lap sponges is strictly controlled to guarantee their safety during surgical procedures. To guarantee that the sponges are free of flaws and impurities, they are created in a sterile setting and put through stringent quality control methods. Additionally, until they are ready for use in the operating room, they are packed in a way that keeps them sterile. The sponges must be disposed away safely and appropriately after being utilised in a surgical operation. To make sure that no sponges were left within the patient, the sponges should be retrieved and counted. After that, they are disposed of in accordance with local laws and standards. To make sure they don't endanger the public's health, this can entail cremation or other sterilisation techniques. The sponges must be disposed away safely and appropriately after being utilised in a surgical operation. To make sure that no sponges were left within the patient, the sponges should be retrieved and counted. After that, they are disposed of in accordance with local laws and standards. To make sure they don't endanger the public's health, this can entail cremation or other sterilisation techniques. A vital instrument for assuring patient safety during surgical operations is sterile x-ray detectable lap sponges. They lessen the possibility of retained sponges and make it simpler for surgical staff to find sponges. These sponges must be used in accordance with strict guidelines to ensure their effectiveness and safety, and they must be disposed of properly. Sterile x-ray detectable lap sponges are beneficial in various medical contexts when wound cleaning and dressing are needed, while being utilised predominantly in surgical settings.
Back to Blog
Using a processor that can process 64-bit data values in a single clock cycle is referred to as 64-bit computing. Older processors, such those used in the early days of computing, could only accept 8-bit or 16-bit data, in comparison. Numerous advantages have resulted from the switch to 64-bit computing, including better security and more processor and memory capacity.
Increased processing power is one of 64-bit computing's key advantages. Processing is quicker and more effective because 64-bit processors can do more computations per clock cycle than their 32-bit equivalents. For operations requiring a lot of data, like video editing or scientific simulations, this is very advantageous. Advantages: 64-bit computing gives more processing capability in addition to more memory capacity. While 64-bit processors may potentially address up to 16 exabytes of memory, 32-bit CPUs are only capable of accessing up to 4GB of RAM. For applications that need a lot of memory, such virtual machines or databases, this enhanced memory capacity is very beneficial. Enhanced security is a benefit of 64-bit computing as well. The security protections found in 64-bit processors make it more difficult for hackers to take advantage of software flaws. For instance, a feature known as "address space layout randomization" (ASLR) on 64-bit processors makes it more difficult to exploit vulnerabilities by assisting attackers in guessing the memory addresses of crucial system components. Disadvantages: 64-bit computing, however, might also have certain drawbacks. Older software may not work with 64-bit CPUs, which is one of the biggest difficulties. For businesses that depend on old software that hasn't been upgraded to work with 64-bit architectures, this might be very difficult. In rare circumstances, it can be required to employ specialised software emulation tools or to run older apps in a virtual environment in order to maintain compatibility. The fact that 64-bit computing may use more resources than 32-bit computing is another possible problem. As a result of their increased data handling capacity, 64-bit processors may need more memory and computing power to function properly. Higher hardware expenses and increased power usage may follow from this. The transition to 64-bit computing is probably going to continue in the upcoming years despite these difficulties. The advantages of 64-bit computing are projected to increase as more applications are created to utilise 64-bit architectures. Additionally, improvements in hardware and software technologies are probably going to alleviate some of the issues with 64-bit computing, making it a more alluring choice for businesses trying to boost their computing power.
Back to Blog
Hydro Chlorofluorocarbon (HCFC) are substances composed of fluorine, chlorine, hydrogen, and carbon. Some of the chemicals in this class are regarded by both the scientific and industrial communities as suitable short-term replacements for chlorofluorocarbons.
In substitution of CFCs, the usage of Hydro Chlorofluorocarbon (HCFC) gained popularity in the 1980s. They were at first thought to be a more eco-friendly option, but it was subsequently found that they still presented a serious damage to the ozone layer. Ozone depletion potential (ODP) for HCFCs varies from 0.01 to 0.1, which is lower than that of CFCs but still puts the ozone layer at danger. According to Coherent Market Insights the Hydro Chlorofluorocarbon (HCFC) Market Global Industry Insights, Trends, Size, Share, Outlook, and Opportunity Analysis, 2022-2028. The goal of the Montreal Protocol, an international agreement agreed in 1987, was to gradually phase out the manufacture and use of compounds that deplete the ozone layer, notably HCFCs. The treaty established deadlines for both industrialised and developing nations to phase out HCFCs, with the ultimate aim of doing so by the year 2030. A number of regulatory strategies, such as lowering production and consumption, establishing licencing and quota systems, and introducing substitute materials and technological advancements are being used to phase out HCFCs. Hydrofluorocarbons (HFCs), which have 0% ODP but are strong greenhouse gases, and natural refrigerants such carbon dioxide, ammonia, and hydrocarbons are the most popular alternatives to HCFCs. Many nations have had success with the phase-out of HCFCs, especially developed ones like North America and Europe. In underdeveloped nations, where HCFCs are still extensively utilised in numerous industries, the phase-out has been slower. This is caused by a number of things, such as the price of alternative technologies, a lack of finance, and a lack of technical competence. The environment and human health are seriously endangered by the continuous usage of HCFCs. The ozone layer being destroyed by HCFCs has been related to higher amounts of UV light reaching the earth's surface. Human health may be negatively impacted by this, increasing the chance of skin cancer, cataracts, and immune system suppression, among other health problems. With global warming potentials (GWPs) that vary from 93 to 4,750 times that of carbon dioxide, HCFCs are likewise powerful greenhouse gases. Organic substances known as HCFCs have been utilised extensively as blowing agents, solvents, and refrigerants. Despite having less of an effect on the ozone layer than CFCs, they nonetheless provide a serious threat to the environment and public health. A number of regulatory strategies, such as lowering production and consumption, establishing licencing and quota systems, and introducing substitute materials and technological advancements are being used to phase out HCFCs. To preserve the ozone layer and lessen the effects of climate change, HCFCs must be phased out successfully. |
 RSS Feed
RSS Feed