ArchivesCategories |
Back to Blog
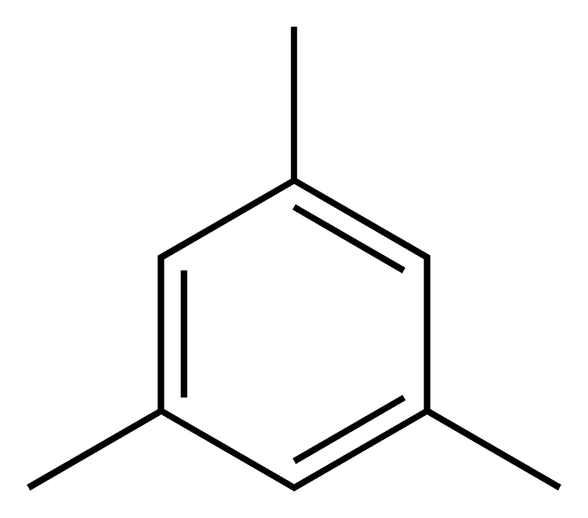
Mesitylene, commonly known as 1, 3, 5-trimethylbenzene, has the chemical formula C6H3 (CH3)3. It is an odourless liquid with a recognisable fragrant scent. Mesitylene belongs to the group of substances known as aromatic hydrocarbons, which are distinguished by their strong fragrance and cyclic arrangement of carbon atoms.
By alkylating benzene with propylene, mesitylene is made commercially. Small quantities of it are also naturally present in some kinds of crude oil and coal tar. The melting point of mesitylene is -45°C and its boiling point is 166°C. Although it is insoluble in water, several organic solvents, including benzene, toluene, and chloroform, can dissolve it. Mesitylene has a high degree of symmetry, which is one of its most prominent characteristics. With regard to its three methyl groups and benzene ring, the molecule is symmetrical. Mesitylene has several intriguing physical characteristics as a result of its symmetry, such as the capacity to crystallise with a high degree of order. Mesitylene is frequently employed as a solvent in organic chemistry, especially in processes involving delicate substances or high temperatures. It is a great option for high-temperature reactions due to its low vapour pressure and comparatively high boiling point. Mesitylene is also utilised as a raw material to make other organic compounds, such medications and colours. Mesitylene is used as a fundamental component in the synthesis of metal-organic frameworks (MOFs), which is a fascinating application. A type of materials known as MOFs is made up of metal ions or clusters that are joined by organic ligands. These substances have a broad variety of possible uses, such as gas storage, sensors, and catalysts. Because of its capacity to bind with metal ions, mesitylene can be employed as a ligand in the production of MOFs. Mesitylene may be used as a gasoline additive, according to other research. Gasoline's octane rating, a gauge of a fuel's capacity to withstand knocking or explosion, can be raised by adding mesitylene. Increasing the octane rating is a crucial objective for petrol makers since knocking may result in engine damage and decreased performance. Mesitylene is an appealing option for a gasoline additive since it is a very inexpensive and plentiful substance. Despite having several beneficial uses, mesitylene does have some disadvantages. Its potential toxicity as well as its flammability make it a dangerous substance. Mesitylene exposure can irritate the skin, eyes, and respiratory system. Although additional study is required to properly grasp its possible health consequences, it is also thought to be a probable carcinogen. Mesitylene is a valuable and adaptable substance having several uses in organic chemistry, materials science, and fuel technology. It is a desirable option for a variety of applications due to its high degree of symmetry and comparatively low toxicity. To guarantee its safe usage, mesitylene must be handled carefully and cautiously, just like any other chemicals.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed