ArchivesCategories |
Back to Blog
Based on the chemical and physical characteristics of the components of a mixture, liquid chromatography (LC) is a separation technique used to separate and analyse the components. It is extensively employed in many different industries, including biology, medicines, and environmental monitoring. In liquid chromatography, a sample is dissolved in a solvent before being passed through a stationary phase, where separation takes place as a result of the components of the sample interacting differently with the stationary phase.
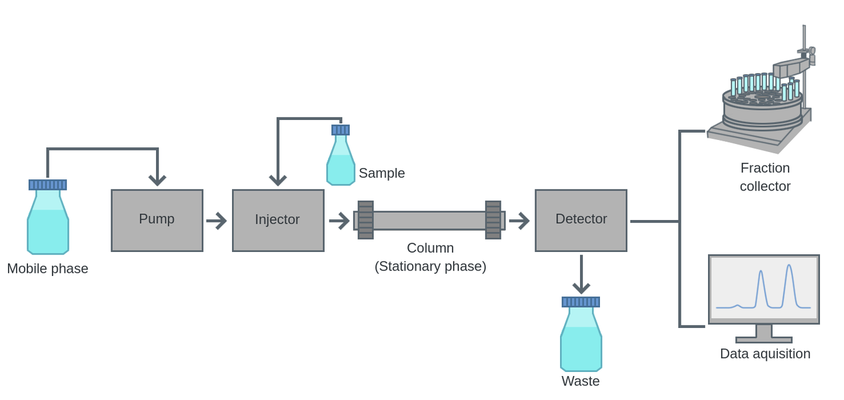
High-performance liquid chromatography (HPLC), ion chromatography, size exclusion chromatography, and affinity chromatography are a few examples of diverse liquid chromatography methods. The kind of sample and the necessary separation criteria determine which approach is used. The most used kind of liquid chromatography is HPLC. The sample is delivered to the stationary phase, which is commonly a column filled with silica-based particles, using a high-pressure pump. The difference in how the sample components interact with the stationary phase leads to the separation, with some components remaining in the column longer than others. Afterward, the separated components are detected and measured using a detector like a mass spectrometer or a UV-Vis spectrophotometer. Ion chromatography is another type of liquid chromatography that is used to separate ions based on their charge and affinity to the stationary phase. It is commonly used in the analysis of inorganic ions such as chloride, nitrate, and sulfate in water samples. Using size exclusion chromatography, molecules are separated according to size. Smaller molecules can enter the pores of the porous beads that make up the stationary phase, while bigger molecules are blocked from doing so and elute first. The purification of biomolecules like proteins and nucleic acids frequently uses this method. According to a molecule's affinity to a particular ligand that is immobilised on the stationary phase, affinity chromatography is used to separate the molecules. Using a particular ligand to bind to the target protein, this approach is frequently used to purify proteins. The choice of stationary phase and mobile phase, as well as the separation factors like column temperature, flow rate, and gradient profile, all affect the outcome of liquid chromatography. By using reversed-phase chromatography, where a hydrophobic stationary phase is utilised to separate hydrophobic molecules, the stationary phase may be altered to improve the separation of certain components. Liquid chromatography has experienced major technological and instrumentation developments in recent years. For instance, finer resolution and quicker separation times have been made possible by the advent of ultra-high performance liquid chromatography (UHPLC). UHPLC offers a better efficiency and quicker separation times because it employs smaller particle sizes in the stationary phase and higher pressures throughout the system. Mass spectrometry being used as a detector in liquid chromatography is another development. In comparison to conventional UV-Vis detection, mass spectrometry offers a more focused and sensitive detection approach. Liquid chromatography-mass spectrometry (LC-MS), a technique that combines liquid chromatography with mass spectrometry, is frequently used to analyse tiny molecules, biomolecules, and metabolites. Liquid chromatography is a flexible separation method with several applications in a variety of industries. The kind of sample and the necessary separation criteria determine which approach is used. Because of improvements in apparatus and technology, liquid chromatography is now a crucial tool in contemporary analytical chemistry. These improvements include greater resolution, quicker separation periods, and more sensitive detection techniques.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed